Photoelectric effect
A metal surface loses negative charge due to the ejection of electrons from its surface when exposed to UV light.
It is the phenomenon of emission of the electrons from the metal surfaces when the radiations of suitable frequency is made to fall on these surfaces.
The electrons so emitted from the metal surfaces are called photoelectrons and the electric current constituted by these electrons is called photoelectric current. About all the metals, when exposed to UV radiations emit photoelectrons but the alkali metals like, Na, K, Rb, and Cs emit photoelectrons on their exposure to the visible light as the alkali metals have low work function. However, the photoelectric effect, in metals like Zn, Cd, Mg etc. is observed, when are exposed to heavy ultraviolet radiations.
experimental study of photoelectric effect
Photoelectric current was extensively studied by Lenard and R. A. Millikan in 1900, using the experimental set up as given in Fig. 11.02.

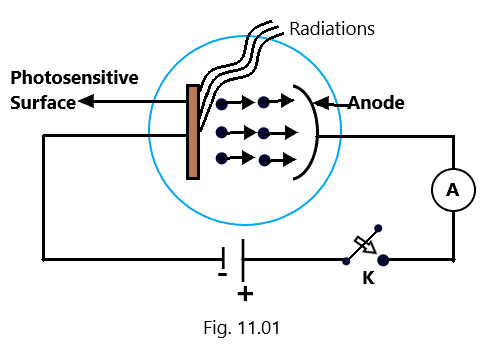
The experimental set up to study the photoelectric effect consists of an evacuated glass tube fitted with two metallic electrodes A and C and a quartz window (W) is also sealed with the evacuated tube to facilitate the smooth entrance of UV light radiations. When the monochromatic light radiations of suitable frequency coming from a source S falls on the photosensitive plate C, made as a cathode, the emission of photoelectrons starts taking place at the cathode in the form of photosensitive material and these photoelectrons emitted at cathode are collected by the anode. (Photoelectric effect)
The two electrodes C and A are connected to a H.T. battery, a potential divider arrangement, and a milliammeter. The amount of potential difference between the electrodes C and A can be varied using Rh and measured by voltmeter. The polarity of potential difference between C and A can be changed with the help of commutator and so a desired positive or negative potential can be maintained at A w.r.t. the cathode (plate C). (Photoelectric effect)
The flow of photoelectric current due to the emission of photoelectrons can be measured by a millimeter (mA) connected in the circuit while the potential difference between the plates can be measured with the help of voltmeter. Changing the magnitude and sign of the anode potential w.r.t. the cathode, the corresponding values of the photoelectric current can noted. Using this experimental set up, the photoelectric effect can be studied in terms of the variation of photoelectric current with (a) with the intensity of the incident radiations (b) the potential difference between plate C and A (c) the frequency of the incident radiations and (d) the nature of the material of plate C. (Photoelectric effect)
1. Effect of Intensity of light on photoelectric current:
Let us consider that the collector plate A is held at some desired positive potential with respect to the emitter C (photosensitive material). The photoelectrons emitted from the cathode emitter, they will be accelerated towards the collector anode C. If the radiations of some suitable fixed frequency fall on the electrode C at a fixed suitable potential difference between two electrodes, then the photoelectric current increases proportionally with the intensity of incident radiations as shown in [Fig. 11.03].

The intensity of the incident light falling on the cathode made of photosensitive material, is gradually increased and the corresponding photoelectric current is recorded each time. If a graph is plotted between the intensity of the light radiation and corresponding values of photoelectric current, we get a straight line, showing that the number of photoelectrons emitted per second is directly proportional to the intensity of light. (Photoelectric effect)
2. Effect of Potential on photoelectric current:
Keeping the anode A at zero potential initially and the light of some frequency greater than its threshold value for the cathode material and some fixed value of intensity (I1) is made to fall on the plate C (cathode) made of the photosensitive material. It is observed that some current (=OK) is noted even the accelerating potential applied on the anode A is zero because of the reason that some energetic photoelectrons reach the anode by themselves. (Photoelectric effect)
When the positive potential at anode A is gradually increased and corresponding value of photoelectric current is recorded each time, it is found that the photoelectric current increases as the accelerating potential increases till a stage is attained when all the photoelectrons emitted from cathode C reach the anode A and the photoelectric current increases no more with the increase in the accelerating potential. This maximum value of the photoelectric current is called Saturation Current, which corresponds to the state when all the photoelectrons emitted by the plate C are attracted at the collector/anode plate A. (Photoelectric effect)
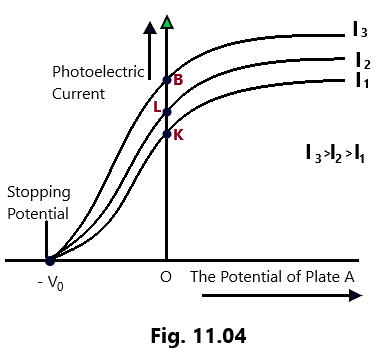
Now, we apply negative potential to the collector/anode plate A, with respect to the cathode plate C. A decrease in the photoelectric current will be seen now due to the reason that the photoelectrons emitted by the cathode get deaccelerated due the negative potential at A. The gradual increase in the negative potential at A, will cause fewer and fewer photoelectrons to reach the anode and results in the gradual decrease in photoelectric current until it becomes zero corresponding to a certain value of negative potential at plate A. Even the fastest photoelectrons with maximum kinetic energy cannot reach the anode collector plate A. (Photoelectric effect)
The least value of negative potential (V0) applied to the anode A, w.r.t. cathode C, corresponding to which all the photoelectrons emitted from cathode C are stopped resulting the photoelectric current to become zero, is called as Stopping Potential or Cut off Potential.
It is evident from the above discussion that no photoelectron reach the anode at the stopping potential V0 and hence, the work done by the stopping potential in doing so, must be equal to the maximum kinetic energy of the photoelectrons, therefore,
Kmax = eV0 = (1/2) m v2max
where, e= charge of each photoelectrons, m= mass of each photoelectrons and vmax = the maximum velocity of photoelectron. The experiment is repeated whit higher intensities of I2 and I3 of the light radiations incident on the cathode as the electron emitter keeping the frequency of the incident radiations constant at all the times. It is observed that the saturation current increases with an increase in the intensity of the incident radiations, while the stopping potential remains the same and is independent of the intensity of the incident radiations. The following conclusions are drawn on the basis of the above discussion.
(1) The kinetic energy possessed by all the photoelectrons is not the same.
(2) The value of stopping potential is independent of the intensity of radiation for the same material of the photosensitive plate (cathode C) and same frequency.
3. Effect of Frequency of Incident Radiations on Photoelectric Current:
The effect of frequency of incident radiations on the photoelectric effect in terms of photoelectric current is studied by keeping the intensity of radiations fixed but the frequency of incident radiations is varied so that the saturation current remains constant in each case. (Photoelectric effect)

The potential on the anode plate A is gradually reduced to zero at some fixed frequency (say v1), and then increased in negative direction till the stopping potential is reached. On repeating the the experiment for the different values of the frequencies of incident radiations, the plot of the graphs come as shown in [Fig.11.05]. The following conclusions are drawn on the basis of the above discussion. (Photoelectric effect)
(i) The stopping potential is different for the radiations of different frequencies. It shows that the stopping potential is a function of the frequency of the incident radiations and independent of its intensity. (Photoelectric effect)
(ii) The value of stopping potential increases with increase in the frequency of the incident radiations. (Photoelectric effect)
(iii) Larger the value of the frequency of the incident radiations, larger will be the maximum kinetic energy of the emitted photoelectrons and so, larger will be the retarding potential needed to prevent the arrival of such photoelectrons completely to the collector plate A.
(iv) The saturation current is independent of the frequency of the incident radiation but it strongly depends on the intensity of the incident radiation.
4. Effect of the Nature of the Materials of the Plates:
Let us suppose that A and B are the two metals of different types and a graph is drawn between the frequency of the incident radiations and the corresponding values of the stopping potential for these two different metals that comes the same as shown in[Fig. 11.06] (Photoelectric effect)

(1) The stopping potential increases linearly with the frequency of the incident radiations for any photosensitive material.
(2) There is a certain minimum cut off frequency called as threshold frequency corresponding to which the stopping potential is zero for any given photosensitive material. (Photoelectric effect)
(3) The graphs are straight lines parallel to each other having same slope two different metals A and B, but the threshold frequencies are different for the two metals. (Photoelectric effect)
Thus, there exists a certain incident radiation, for a given photosensitive material, for which the photoelectric emission is just possible and below this frequency, the photoelectric emission can not be thought of to take place. This frequency is called as threshold frequency. The photoelectric emission is an instantaneous process and the time lag between the incidence of radiation and the emission of photoelectron is less than 10-9 sec. (Photoelectric effect)
laws of photoelectric emission
Lenard and Millikan, on the basis of the results and observations of their experiments related to the photoelectric effect, put forward some laws which are called laws of photoelectric effect, describe as follows. (Photoelectric effect)
(i) There exists a certain minimum cut off frequency for a given photosensitive material, below which the photoelectric emission is not possible, irrespective of the intensity of the incident radiation. This particular frequency is called as threshold frequency.
(ii) The photoelectric current is directly proportional to the intensity of the incident radiations, under the condition that the frequency of incident radiations is greater than the threshold frequency.
(iii) The maximum kinetic energy of the emitted photoelectrons, above the threshold frequency, is independent of the intensity of incident radiations but depends upon the frequency of the radiations.
(iv) The photoelectric emission is an instantaneous process. and the time lag between the incidence of radiations and the emission of photoelectrons is very small, less than 10-9 sec.
Einstein’s photoelectric equation
Albert Einstein, provided a complete and successful explanations to the laws of photoelectric emission on the basis of plank’s quantum theory of light radiations. According to Plank’s quantum theory, the light radiations are considered to consist of tiny packets of energy or energy particles called as quanta. Each quantum of energy is called photon. The energy of each photon is given by E = hv – – – – (1) where, h is the Plank’s constant and v is the frequency of the radiation quantum. As the photon is an energy packet so it travels with a speed equal to that of light.`(Photoelectric effect)
Einstein’s explanations to the laws of photoelectric emission is based on the assumption that one photon emits only one photoelectron from the photosensitive material if it is capable of doing so (photon must have suitable energy)(Photoelectric effect)

Let us suppose that a photon of energy, E = hv is made to strike the photosensitive metal plate as shown in [Fig. 11.07] give above. As a result, the photon will interact with the electron of the target photosensitive metal plate through elastic collision. The energy of the photon will be used in two ways.
(i) A part of the energy of the photon is used to overcome the work function (W0) of the metal to liberate the electron from the interior of it and bring to its surface.
(ii) Rest of the energy of the photon is used to provide the kinetic energy to the emitted photoelectron at the surface. Thus, maximum kinetic energy gained by the ejected photoelectron, Kmax = (1/2) m v2max . Applying conservation law of energy,
hv = work function + Kmax = W0 + Kmax = W0 + (1/2) m v2max => (1/2) m v2max = hv – W0 – – – – – (2)
When v0 is the threshold frequency of the incident light photon so that its energy (hv0) is sufficient to just eject the photoelectron on the metal surface without imparting any kinetic energy to it. In this particular case, the energy of the photon (hv0) will be equal to the work function of the metal, thus, hv0 = W0, with the substitution of this eqn. in eqn. (2), we get (1/2) m v2max = hv – hv0 (1/2) m v2max = h(v – v0) – – – -(3) These equations (2) and (3) are called as photoelectric equations.
Explanations of the laws of photoelectric emission
(1) Let us consider that the frequency of the incident light photon is less than the threshold frequency (v < v0), then the kinetic energy of the emitted photon, from eqn. (3), will be negative. But the kinetic energy can never be negative. Thus, the photoelectric emission can never be thought of to take place if the frequency of the incident light photon is less than the threshold frequency. This is the first law of photoelectric emission.
(2) According to Einstein theory, the photoelectric effect comes into existence when a single photon is absorbed by a single electron. Therefore, it is a one photon- one electron phenomenon. Thus, the number of photoelectrons emitted by the photosensitive metal plate would be large when an intense beam of radiation is incident, because of the reason that the intensity of radiation is proportional to the number of photons per unit area, per unit time.
Therefore, an intense radiation will contain a large number of photons and so, the number of photoelectrons emitted will also be large. It proves that the number of photoelectrons emitted is a function of the intensity of incident radiations. Further, there is no effect of frequency of the incident radiation on the number of photoelectrons emitted. It is because one proton is capable of ejecting only one electron, provided frequency is greater than the threshold frequency. This is the second law of photoelectric emission.
(3) From the equation (3), it is clear that the value of maximum kinetic energy of the emitted photoelectrons depends linearly on the frequency of the incident radiations. Therefore, it is adequate to prove that the maximum kinetic energy of the emitted photoelectrons increases with the increase in the frequency of the incident radiations. Since, the Einstein’s equation does not involve a factor representing intensity of the incident radiations. It proves that the maximum kinetic energy of the emitted photoelectrons is independent of the intensity of the incident radiations. This is the first law of photoelectric emission.
(4) According to Einstein’s theory, the basic process involved in photoelectric emission is the absorption of a photon of light by an electron through the elastic collision. Therefore, as the photon is absorbed, the emission of the photoelectron takes place instantaneously. It may be noted that irrespective of the intensity of the incident radiation, the photoelectric emission is instantaneous.
